Preclinical Research And Regulatory Processes In Drug Development Panel Was Held
The "Panel on Preclinical Research and Regulatory Processes in Drug Development" moderated by Prof. Dr. Uygar Halis TAZEBAY, a researcher at Turkiye Cancer Institute, started with the opening speech of Prof. Dr. Hakan EROĞLU, President of TÜSEB.
Starting his opening speech by mentioning the importance of the pharmaceutical industry in the health sector, Prof. Dr. Hakan EROĞLU emphasized the importance of supporting R&D-oriented innovative initiatives and following the advances in the pharmaceutical industry, and mentioned the activities carried out by TÜSEB to encourage and support pre-clinical and post-clinical studies in drug development processes. Prof. Dr. EROĞLU also stated that every step taken to contribute to the domestic and national transformation in the pharmaceutical industry and to reduce our country's foreign dependence in this field is valuable, and touched upon the roles undertaken by TÜSEB in this direction.
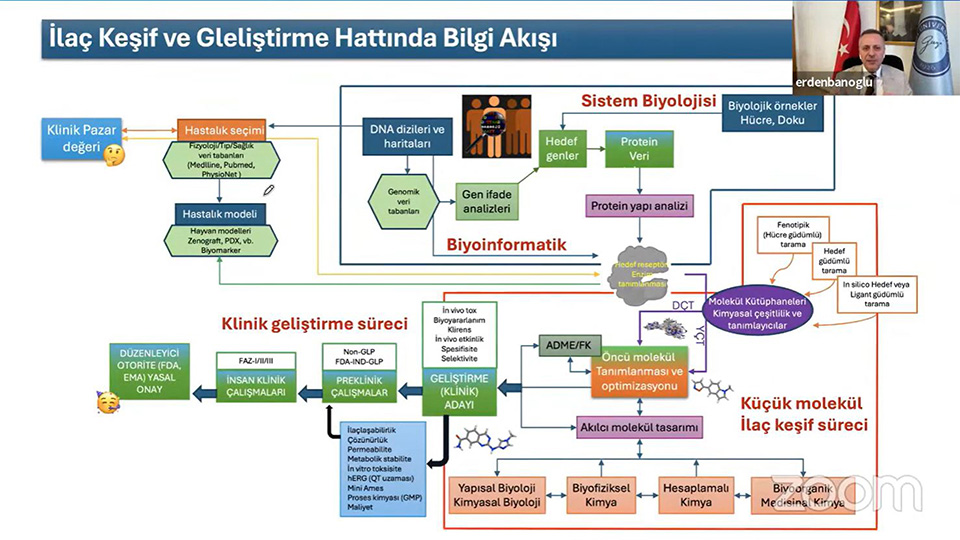
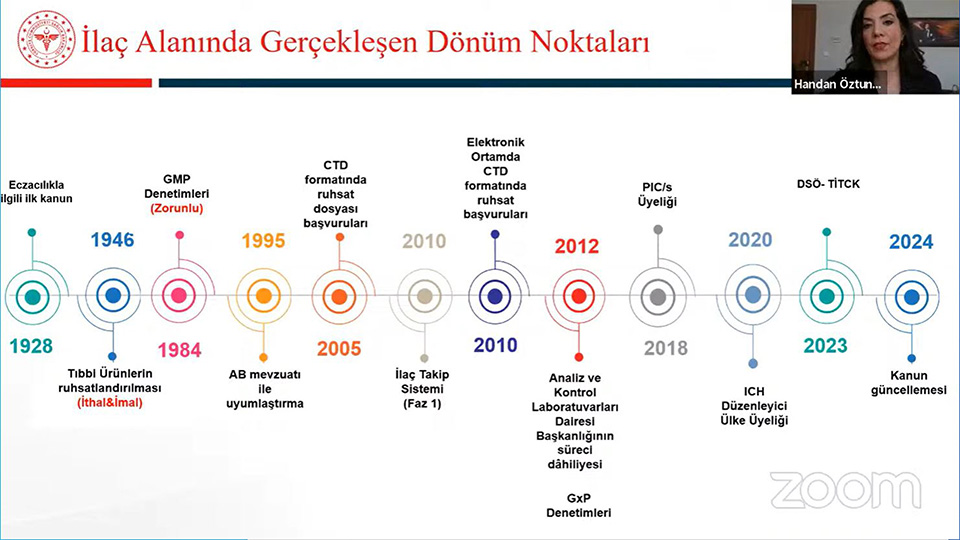
In the panel where detailed and up-to-date information on preclinical research and licensing processes in drug development were shared, Prof. Dr. Erden BANOĞLU, Dean of Gazi University Faculty of Pharmacy, made a presentation titled "Preclinical Research on the Road from Molecule to Drug", and Uzm. Pharm. F. Handan ÖZTUNCA, Head of the Drug Licensing Department of the Turkish Medicines and Medical Devices Agency (TİTCK), made a presentation titled 'Drug Licensing Processes and the Reflection of Preclinical Study Results on the Processes'.
As TÜSEB, we will continue to establish scientific platforms that will bring together all relevant stakeholders in the field of health, which is of strategic importance for our country, in order to lead the development of health sciences and technologies in our country.
Click here to watch the panel, which was broadcast live on TÜSEB's Official YouTube Channel.